Introduction
Discrete morphological models are not only useful for tree estimation, as was done in Tutorial Discrete morphology - Tree Inference, but also to ask specific questions about the evolution of the morphological character of interest. Specifically, there are a few types of analyses that we might be interest in. First, we can test different models of morphological evolution, such as reversible and irreversible models, and estimate rates under these models. For example, using an irreversible model of evolution, we can test, for example, for Dollo’s law of a complex character that can be lost but not gained again (Goldberg and Igić 2008).
Additionally, we might be interest in ancestral state estimation, or mapping transition on the phylogeny. Commonly the central problem in statistical phylogenetics concerns marginalizing over all unobserved character histories that evolved along the branches of a given phylogenetic tree according to some model, $M$, under some parameters, $\theta$. This marginalization yields the probability of observing the tip states, $X_\text{tip}$, given the model and its parameters, $P( X_\text{tip} | \theta, M ) = \sum_{X_\text{internal}} P( X_\text{internal}, X_\text{tip} \mid \theta, M )$. One might also wish to find the probability distribution of ancestral state configurations that are consistent with the tip state distribution, $P( X_\text{internal} \mid X_\text{tip}, \theta, M )$, and to sample ancestral states from that distribution. This procedure is known as ancestral state estimation.
%Finally, we might be interested in testing for correlated evolution between discrete morphological characters. %For example, % This tutorial will provide a discussion of modeling morphological characters and ancestral state estimation, and will demonstrate how to perform such Bayesian phylogenetic analysis using RevBayes (Höhna et al. 2016).
The data
Create a directory on your computer for this tutorial. In this directory, create a subdirectory called data, and download the data files that you can find on the left of this page.
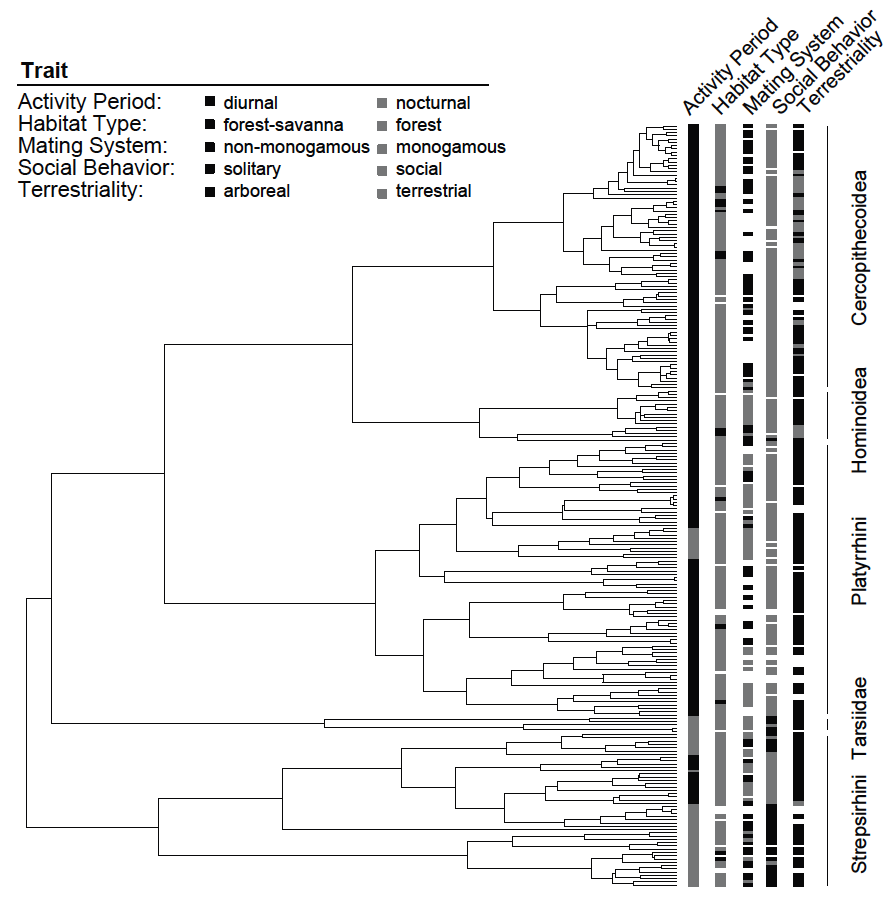
We have taken the phylogeny from Magnuson-Ford and Otto (2012), who took it from Vos and Mooers (2006) and then randomly resolved the polytomies using the method of Kuhn et al. (2011) and the trait data from Redding et al. (2010). In the data folder, you should now have the following files:
- primates_tree.nex: Dated primate phylogeny including 233 out of 367 species.
- primates_activity_period.nex:
A file with the coded character states for primate species activity time. This character has just two states:
0= diurnal and1= nocturnal. - primates_habitat.nex:
A file with the coded character states for primate species habitay. This character has just two states:
0= forest and1= savanna. - primates_solitariness.nex:
A file with the coded character states for primate species social system type. This character has just two states:
0= group living and1= solitary. - primates_terrestrially.nex:
A file with the coded character states for primate species terrestrially. This character has just two states:
0= arboreal and1= terrestrial. - primates_males.nex:
A file with the coded character states for the number of males within a group per primate species. This character has four states:
0= single male,1= single and multi male, and2= multi male. - primates_mating_system.nex:
A file with the coded character states for primate species mating-system type. This character has four states:
0= monogamy,1= polygyny,2= polygynandry, and3= polyandry. - primates_diet.nex:
A file with the coded character states for primate species diet. This character has six states:
0= frugivore,1= insectivore,2= folivore,3= gummnivore,4= omnivore, and5= gramnivore.
Scripts
For more complex models and analyses, it’s useful to create separate Rev scripts that contain all the model parameters, moves, and functions for different model components (e.g. the substitution model and the clock model).
Create another subdirectory called scripts.
In this tutorial, you will work primarily in your text editor and create a script file that can be easily managed and modified. Examples of all the commands used to perform each analysis are also provided at the top of this page under Scripts but try to write the complete scripts yourself from the beginning to ensure you understand all the steps involved and the differences between setting up each analysis.
Exercises
Click on the first exercise to begin!
- Goldberg E.E., Igić B. 2008. On Phylogenetic Tests of Irreversible Evolution. Evolution. 62:2727–2741. 10.1111/j.1558-5646.2008.00505.x
- Höhna S., Landis M.J., Heath T.A., Boussau B., Lartillot N., Moore B.R., Huelsenbeck J.P., Ronquist F. 2016. RevBayes: Bayesian Phylogenetic Inference Using Graphical Models and an Interactive Model-Specification Language. Systematic Biology. 65:726–736. 10.1093/sysbio/syw021
- Kuhn T.S., Mooers A.Ø., Thomas G.H. 2011. A Simple Polytomy Resolver for Dated Phylogenies. Methods in Ecology and Evolution. 2:427–436. 10.1111/j.2041-210X.2011.00103.x
- Magnuson-Ford K., Otto S.P. 2012. Linking the Investigations of Character Evolution and Species Diversification. The American Naturalist. 180:225–245. 10.1086/666649
- Redding D.W., DeWolff C.V., Mooers A.Ø. 2010. Evolutionary Distinctiveness, Threat Status, and Ecological Oddity in Primates. Conservation Biology. 24:1052–1058.
- Vos R.A., Mooers A.Ø. 2006. A New Dated Supertree of the Primates. .